Amarin Corporation Pl.C. (NASDAQ:AMRN) announced today that one of their pending patent applications relating to the ANCHOR indication has received Notice of Allowance from the US Patent and Trademark Office. Meanwhile, changes to the website www.vascepa.com indicate that Vascepa is Now Available. We believe that the company may be issuing a press release as early as tonight or tomorrow to announce the US availability of Vascepa.
ANCHOR Patents
According to the press release, the patent which received notice of allowance is application number 12/815,569 which is titled "Compositions and Methods for Lowering Triglycerides Without Raising LDL-C Levels in a Subject on Concomitant Statin Therapy." The importance of this notice of allowance is that this is the first patent protecting the use of Vascepa in the high trigylceride population which was studied under the ANCHOR clinical trial. The ANCHOR population is 8x - 10x as large as the already approved MARINE trial population of very high triglycerides (>500 mg/dl). This patent, and another pending application, number 13/272,520, both received reasons for allowance in December and the notice of allowance is the next step in the process. In the next week to 10 days, the '520 application should receive notice of allowance as well. As we indicated in December, these ANCHOR patents may be a key factor in any potential buyout for Amarin because the ANCHOR population represents a much larger revenue opportunity if FDA eventually approves Vascepa in this population.
Vascepa Launch
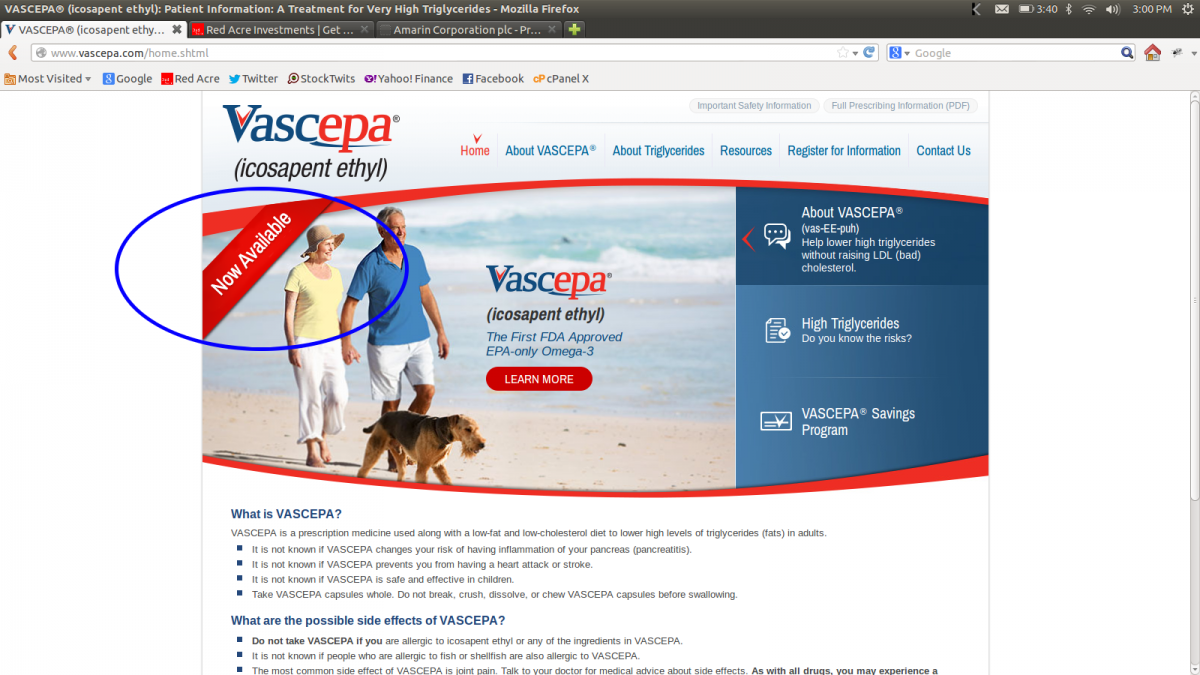
Amarin has previously guided that they planned to launch Vascepa commercially in "early Q1 2013". In preparation for the launch, the company has prepared the website www.vascepa.com. As of today this website indicates that Vascepa is "Now Available" as seen in the screenshot below (click to enlarge).
A visit to Amarin's corporate website still shows Vascepa as "coming soon".
The compnay has also not issued a press release regarding US availability of Vascepa.
Clearly, Vascepa's launch is imminent. We believe the vascepa.com site may be in preparation for the launch, and that the corporate website will be updated once the formal launch announcement is made within the next few days.
A call to AMRN investor relations department was not immediately returned.
In addition to the vascepa.com website, the ompany has launched a website to offer Vascepa savings cards. The saving program involves one free prescription of Vascepa and subsequent refunds, up to $75 such that patients pay no more than $25 per refill. This effectively allows Vascepa to be reimbursed as a Tier II drug even if insurers initially cover the drug at Tier III as long as patients refill for 90 day supplies.
Conclusion
Within the next few days, possibly as soon as this evening, AMRN will issue a press release announcing US availability of Vascepa. We also expect the company to issue a press release regarding the notice of allowance for the '520 patent within the next few weeks. Finally, sometime before the end of February the company should be announcing that they have filed a supplemental New Drug Application for the ANCHOR indication (high triglyceride pateints) for Vascepa.
We remind investors, as we have numerous times in the past, that AMRN is NOT likely to be bought out in the near future. With Vascepa's launch happening now, a deal, if any, is likely to happen much closer to, if not after, the approval of the ANCHOR sNDA.
Depending on how the initial script numbers come in, AMRN stock will either begin a gradual recovery to the $10 - $12 range or drift lower to the $7 - $5 range over the next two months.